Chemotaxis
Collective Behavior of Bacterial Colonies under Growth and Chemotaxis
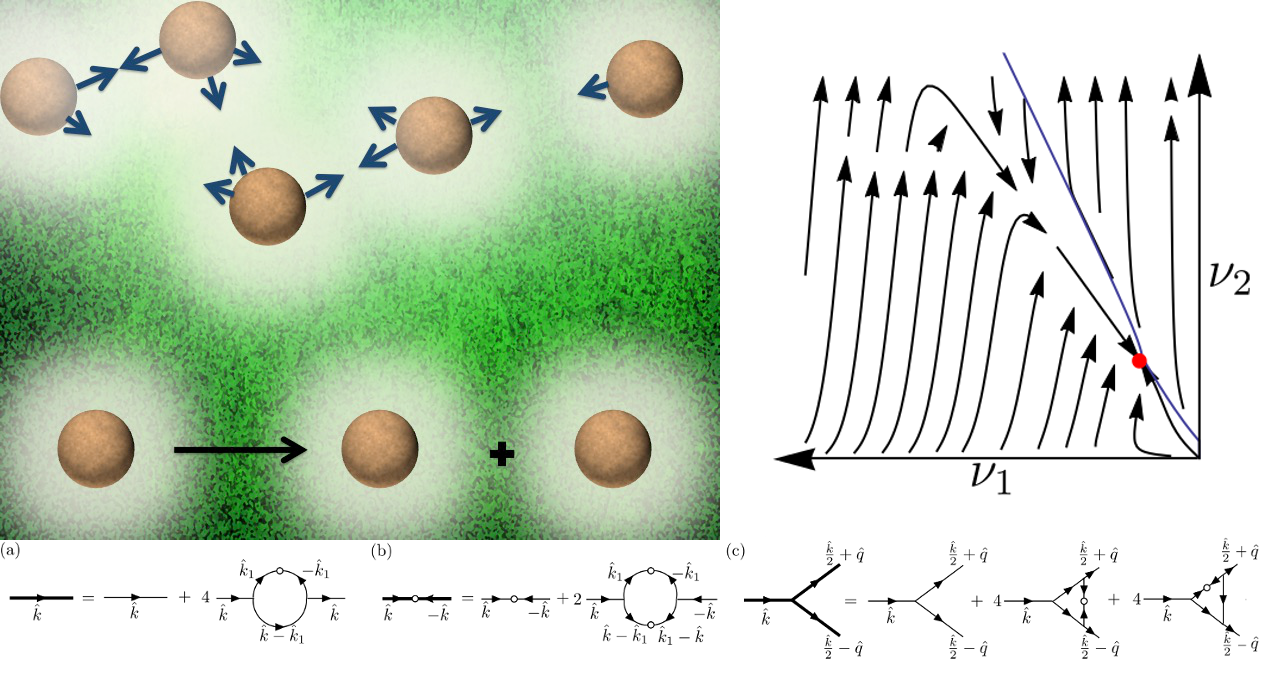
But one of the characteristic features of the long-time dynamics of living cells is that number conservation does not hold due to cell division and death processes, which has consequences on their collective behaviour. We have studied, using dynamical renormalization group methods, the combined effect of this nonequilibrium property of a colony of living cells and long-range chemotactic interactions among the cells. The combination of the effective long-range chemotactic interaction and lack of number conservation leads to a rich variety of phase behaviour in the system, which includes a sharp transition from a phase that is controlled by a weakly coupled perturbatively accessible fixed point to a phase controlled by a nonaccessible strong coupling fixed point.
Group members involved: A. Gelimson and R. Golestanian
Trail guidance of Pseudomonas aeruginosa
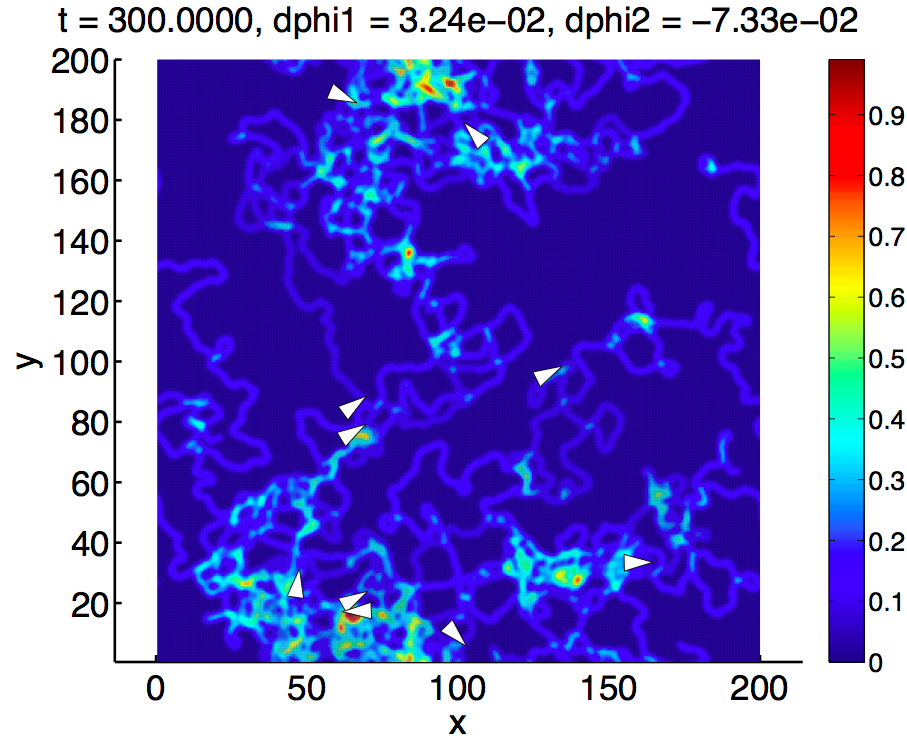
We set up a simple model that captures both the crawling motion and the autochemotactic interaction with the PSL. Based on this model and using numerical and analytical approaches, we study the trail-following behaviour of a bacterium on a preexisting trail and the interaction of a bacterium with its own trail. The latter can lead to an observable dynamics that drastically differs from the underlying microscopic model.
Ultimately, we aim to interpret experimental observations in terms of a simple, physically sound model.
Group members involved: A. Gelimson, T. Kranz and R. Golestanian








